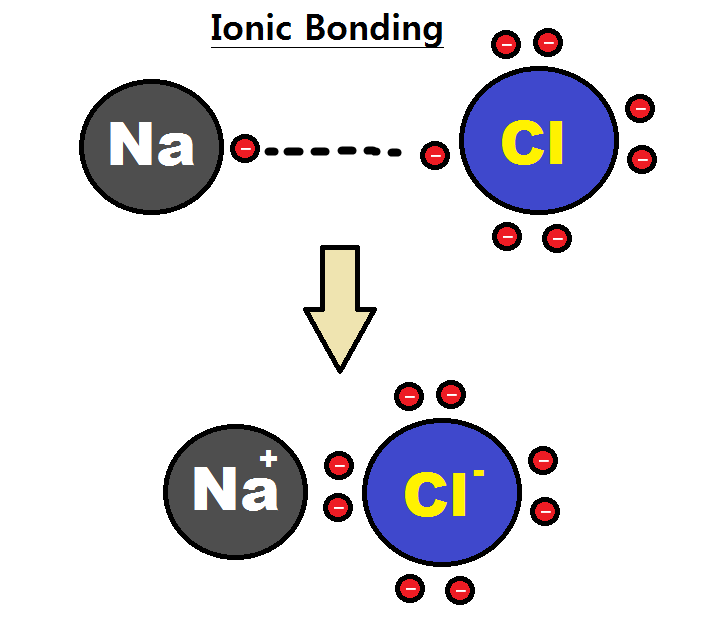
Definition of Ionic Bond
An ionic bond is formed by the complete transfer of electrons from one atom to another. This type of bond gives rise to two oppositely charged ions, one is positive called cation and the other is a negative called anion. It is formed between a metal and a non-metal. Metals can lose electrons and become cations whereas the non-metals can gain electrons and become anions and in this manner, they can achieve noble gas configuration and stabilize their atoms by completing their octet. Hence, in an ionic bond, the metal acts as an electron donor and the non-metal acts as an electron acceptor. The net charge on the compound should be zero in this type of bonding.
Example of Ionic Bond:
Take an example of sodium chloride which is an ionic compound. Sodium (Na) has 11 electrons in its atom and 1 valence electron in its outermost shell which must be donated so that it could be stabilized. On the other hand, Chlorine has 17 electrons in its atom and seven valence electrons in its outermost shell. So, Na donates its one electron to Cl and acquires a positive charge whereas the chlorine accepts that one electron and acquires the negative charge. In this way, the valency of both the atoms is satisfied and they have achieved noble gas configuration.
View More Organic Chemistry Definitions
Related Questions of Organic Chemistry
Draw a mechanism for each of the following transformations: /
Draw and name four terminal alkynes with the molecular formula C6H10.
Propose a stepwise mechanism for the following transformation: /
Write an equation for the proton transfer reaction that occurs when each
Identify the sole product of the following reaction: /
Firefly luciferin is the compound that enables fireflies to glow.
Scorzocreticin (S)-1 was isolated from a plant that is
Draw the condensation product that is expected when each of the following
87. What is the major product of this reaction?
Report the result of the following addition to the proper number of
Show All