Question: A heat engine is operating on a
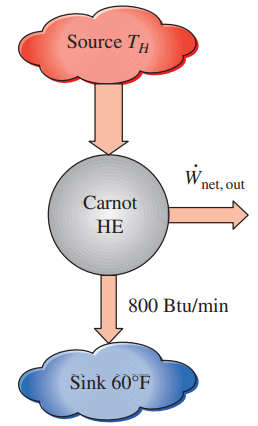
A heat engine is operating on a Carnot cycle and has a thermal efficiency of 47 percent. The waste heat from this engine is rejected to a nearby lake at 60°F at a rate of 800 Btu/min. Determine (a) the power output of the engine and (b) the temperature of the source.
> Repeat Prob. 8–114 for a turbine efficiency of 85 percent. Data from Prob. 8-114: Steam at 3 MPa and 400°C is expanded to 30 kPa in an adiabatic turbine with an isentropic efficiency of 92 percent. Determine the power produced by this turbine, in kW, wh
> Steam at 3 MPa and 400°C is expanded to 30 kPa in an adiabatic turbine with an isentropic efficiency of 92 percent. Determine the power produced by this turbine, in kW, when the mass flow rate is 2 kg/s.
> Steam at 4 MPa and 350°C is expanded in an adiabatic turbine to 120 kPa. What is the isentropic efficiency of this turbine if the steam is exhausted as a saturated vapor?
> Combustion gases enter an adiabatic gas turbine at 1540°F and 120 psia and leave at 60 psia with a low velocity. Treating the combustion gases as air and assuming an isentropic efficiency of 82 percent, determine the work output of the turbine.
> Steam at 100 psia and 650°F is expanded adiabatically in a closed system to 10 psia. Determine the work produced, in Btu/lbm, and the final temperature of steam for an isentropic expansion efficiency of 80 percent.
> Argon gas enters an adiabatic turbine at 800°C and 1.5 MPa at a rate of 80 kg/min and exhausts at 200 kPa. If the power output of the turbine is 370 kW, determine the isentropic efficiency of the turbine.
> The lower half of a 6-m-high cylindrical container is filled with water ( ρ = 1000 kg/m3) and the upper half with oil that has a specific gravity of 0.85. Determine the pressure difference between the top and bottom of the cylinder.
> A piston–cylinder device contains nitrogen gas. During a reversible, adiabatic process, the entropy of the nitrogen will (never, sometimes, always) increase.
> On a T-s diagram, does the actual exit state (state 2) of an adiabatic turbine have to be on the right-hand side of the isentropic exit state (state 2s)? Why?
> Is the isentropic process a suitable model for compressors that are cooled intentionally? Explain.
> Describe the ideal process for an (a) adiabatic turbine, (b) adiabatic compressor, and (c) adiabatic nozzle, and define the isentropic efficiency for each device.
> Reconsider Prob. 8–105. Using appropriate software, investigate the effect of the quality of the steam at the turbine exit on the net-work output. Vary the quality from 0.5 to 1.0, and plot the net-work output as a function of this quality. Data from Pr
> Consider a steam power plant that operates between the pressure limits of 5 MPa and 10 kPa. Steam enters the pump as saturated liquid and leaves the turbine as saturated vapor. Determine the ratio of the work delivered by the turbine to the work consumed
> Water enters the pump of a steam power plant as saturated liquid at 20 kPa at a rate of 45 kg/s and exits at 6 MPa. Neglecting the changes in kinetic and potential energies and assuming the process to be reversible, determine the power input to the pump.
> Liquid water at 120 kPa enters a 7-kW pump where its pressure is raised to 5 MPa. If the elevation difference between the exit and the inlet levels is 10 m, determine the highest mass flow rate of liquid water this pump can handle. Neglect the kinetic en
> Saturated water vapor at 150°C is compressed in a reversible steady-flow device to 1000 kPa while its specific volume remains constant. Determine the work required in kJ/kg.
> Air is compressed isothermally from 13 psia and 55°F to 80 psia in a reversible steady-flow device. Calculate the work required, in Btu/lbm, for this compression.
> Determine the maximum amount of load, in kg, the balloon described in Prob. 2–72 can carry. Data from Prob 2-72: Balloons are often filled with helium gas because it weighs only about one-seventh of what air weighs under identical cond
> Calculate the work produced, in Btu/lbm, for the reversible steady-flow process 1–2 shown in Fig. P8–100E.
> A piston–cylinder device contains helium gas. During a reversible, isothermal process, the entropy of the helium will (never, sometimes, always) increase.
> Does a cycle for which ∮ δQ > 0 violate the Clausius inequality? Why?
> A Carnot refrigerator operates in a room in which the temperature is 25°C. The refrigerator consumes 500 W of power when operating and has a COP of 4.5. Determine (a) the rate of heat removal from the refrigerated space and (b) the temperature of the ref
> A completely reversible refrigerator operates between thermal energy reservoirs at 450 R and 540 R. How many kilowatts of power are required for this device to produce a 15,000-Btu/h cooling effect?
> A Carnot refrigerator absorbs heat from a space at 15°C at a rate of 16,000 kJ/h and rejects heat to a reservoir at 36°C. Determine the COP of the refrigerator, the power input in kW, and the rate of heat rejected to the high-temper
> An air-conditioning system is used to maintain a house at 70°F when the temperature outside is 100°F. The house is gaining heat through the walls and the windows at a rate of 800 Btu/min, and the heat generation rate within the house from people, lights,
> A heat pump operates on a Carnot heat pump cycle with a COP of 12.5. It keeps a space at 24°C by consuming 2.15 kW of power. Determine the temperature of the reservoir from which the heat is absorbed and the heating load provided by the heat pump.
> An air-conditioning system operating on the reversed Carnot cycle is required to transfer heat from a house at a rate of 750 kJ/min to maintain its temperature at 24°C. If the outdoor air temperature is 35°C, determine the power required to operate this
> A completely reversible refrigerator is driven by a 10-kW compressor and operates with thermal energy reservoirs at 250 K and 300 K. Calculate the rate of cooling provided by this refrigerator.
> Reconsider Prob. 2–72. Using appropriate software, investigate the effect of the number of people carried in the balloon on acceleration. Plot the acceleration against the number of people, and discuss the results. Data from Prob 2-72:
> Determine the minimum work per unit of heat transfer from the source reservoir that is required to drive a heat pump with thermal energy reservoirs at 460 K and 535 K.
> A thermodynamicist claims to have developed a heat pump with a COP of 1.7 when operating with thermal energy reservoirs at 273 K and 293 K. Is this claim valid?
> It is well known that the thermal efficiency of heat engines increases as the temperature of the energy source increases. In an attempt to improve the efficiency of a power plant, somebody suggests transferring heat from the available energy source to a
> Is it possible for a heat engine to operate without rejecting any waste heat to a low-temperature reservoir? Explain.
> It is well established that the thermal efficiency of a heat engine increases as the temperature TL at which heat is rejected from the heat engine decreases. In an effort to increase the efficiency of a power plant, somebody suggests refrigerating the co
> In an effort to conserve energy in a heat-engine cycle, somebody suggests incorporating a refrigerator that will absorb some of the waste energy QL and transfer it to the energy source of the heat engine. Is this a smart idea? Explain.
> How can we increase the COP of a Carnot refrigerator?
> A homeowner buys a new refrigerator with no freezer compartment and a deep freezer for the new kitchen. Which of these devices would you expect to have a lower COP? Why?
> A homeowner buys a new refrigerator and a new air conditioner. Which one of these devices would you expect to have a higher COP? Why?
> What is the highest COP that a refrigerator operating between temperature levels TL and TH can have?
> Balloons are often filled with helium gas because it weighs only about one-seventh of what air weighs under identical conditions. The buoyancy force, which can be expressed as Fb = ρairgVballoon, will push the balloon upward. If the balloon ha
> It is claimed that the efficiency of a completely reversible heat engine can be doubled by doubling the temperature of the energy source. Justify the validity of this claim.
> In tropical climates, the water near the surface of the ocean remains warm throughout the year as a result of solar energy absorption. In the deeper parts of the ocean, however, the water remains at a relatively low temperature since the sunâ€
> An experimentalist claims that, based on his measurements, a heat engine receives 300 Btu of heat from a source of 900 R, converts 160 Btu of it to work, and rejects the rest as waste heat to a sink at 540 R. Are these measurements reasonable? Why?
> Reconsider Prob. 7–79. Using appropriate software, study the effects of the temperatures of the heat source and the heat sink on the power produced and the cycle thermal efficiency. Let the source temperature vary from 300 to 1000°C, and the sink tempera
> What is the Kelvin–Planck expression of the second law of thermodynamics?
> A heat engine operates between a source at 477°C and a sink at 25°C. If heat is supplied to the heat engine at a steady rate of 65,000 kJ/min, determine the maximum power output of this heat engine.
> A Carnot heat engine operates between a source at 1000 K and a sink at 300 K. If the heat engine is supplied with heat at a rate of 800 kJ/min, determine (a) the thermal efficiency and (b) the power output of this heat engine.
> An inventor claims to have developed a heat engine that receives 700 kJ of heat from a source at 500 K and produces 300 kJ of net work while rejecting the waste heat to a sink at 290 K. Is this a reasonable claim? Why?
> A completely reversible heat engine operates with a source at 1500 R and a sink at 500 R. At what rate must heat be supplied to this engine, in Btu/h, for it to produce 5 hp of power?
> An air-conditioning system requires a 35-m-long section of 15-cm-diameter ductwork to be laid underwater. Determine the upward force the water will exert on the duct. Take the densities of air and water to be 1.3 kg/m3 and 1000 kg/m3, respectively.
> A thermodynamicist claims to have developed a heat engine with 50 percent thermal efficiency when operating with thermal energy reservoirs at 1260 R and 510 R. Is this claim valid?
> Reconsider Prob. 7–72. You also know that the metallurgical temperature limit for the blades in the turbine is 1000 K before they will incur excessive creep. Now what is the maximum efficiency for this plant? Data from Prob. 7-72: You are an engineer in
> You are an engineer in an electric-generation station. You know that the flames in the boiler reach a temperature of 1200 K and that cooling water at 300 K is available from a nearby river. What is the maximum efficiency your plant will ever achieve?
> Consider two actual power plants operating with solar energy. Energy is supplied to one plant from a solar pond at 80°C and to the other from concentrating collectors that raise the water temperature to 600°C. Which of these power plants will have a high
> Is there any way to increase the efficiency of a Carnot heat engine other than by increasing TH or decreasing TL?
> What are the characteristics of all heat engines?
> Somebody claims to have developed a new reversible heat-engine cycle that has the same theoretical efficiency as the Carnot cycle operating between the same temperature limits. Is this a reasonable claim?
> Somebody claims to have developed a new reversible heat-engine cycle that has a higher theoretical efficiency than the Carnot cycle operating between the same temperature limits. How do you evaluate this claim?
> Is it possible to develop (a) an actual and (b) a reversible heat-engine cycle that is more efficient than a Carnot cycle operating between the same temperature limits? Explain.
> What are the two statements known as the Carnot principles?
> The force generated by a spring is given by F = kx, where k is the spring constant and x is the deflection of the spring. The spring of Fig. P2–70 has a spring constant of 8 kN/cm. The pressures are P1 = 5000 kPa, P2 = 10,000 kPa, and P
> Can there be any heat transfer between two bodies that are at the same temperature but at different pressures?
> What are the four processes that make up the Carnot cycle?
> Is a reversible expansion or compression process necessarily quasi-equilibrium? Is a quasi-equilibrium expansion or compression process necessarily reversible? Explain.
> Why does a non-quasi-equilibrium expansion process deliver less work than the corresponding quasi-equilibrium one?
> Why does a non-quasi-equilibrium compression process require a larger work input than the corresponding quasi-equilibrium one?
> Show that processes that use work for mixing are irreversible by considering an adiabatic system whose contents are stirred by turning a paddle wheel inside the system (e.g., stirring a cake mix with an electric mixer).
> Show that processes involving rapid chemical reactions are irreversible by considering the combustion of a natural gas (e.g., methane) and air mixture in a rigid container.
> Consider the process of baking potatoes in a conventional oven. Can the hot air in the oven be treated as a thermal energy reservoir? Explain.
> How do you distinguish between internal and external irreversibilities?
> A block slides down an inclined plane with friction and no restraining force. Is this process reversible or irreversible? Justify your answer.
> A cold canned drink is left in a warmer room where its temperature rises as a result of heat transfer. Is this a reversible process? Explain.
> A vertical piston–cylinder device contains a gas at a pressure of 100 kPa. The piston has a mass of 10 kg and a diameter of 14 cm. Pressure of the gas is to be increased by placing some weights on the piston. Determine the local atmosph
> Why are engineers interested in reversible processes even though they can never be achieved?
> Refrigerant-134a enters the condenser of a residential heat pump at 800 kPa and 35°C at a rate of 0.018 kg/s and leaves at 800 kPa as a saturated liquid. If the compressor consumes 1.2 kW of power, determine (a) the COP of the heat pump and (b
> A house that was heated by electric resistance heaters consumed 1200 kWh of electric energy in a winter month. If this house were heated instead by a heat pump that has an average COP of 2.4, determine how much money the homeowner would have saved that m
> Consider an office room that is being cooled adequately by a 12,000 Btu/h window air conditioner. Now it is decided to convert this room into a computer room by installing several computers, terminals, and printers with a total rated power of 8.4 kW. The
> A household refrigerator runs one-fourth of the time and removes heat from the food compartment at an average rate of 800 kJ/h. If the COP of the refrigerator is 2.2, determine the power the refrigerator draws when running.
> A refrigerator is used to cool water from 23 to 5°C in a continuous manner. The heat rejected in the condenser is 570 kJ/min and the power is 2.65 kW. Determine the rate at which water is cooled in L/min and the COP of the refrigerator. The specific heat
> Water enters an ice machine at 55°F and leaves as ice at 25°F. If the COP of the ice machine is 2.4 during this operation, determine the required power input for an ice production rate of 28 lbm/h. (169 Btu of energy needs to be removed from each lbm of
> An experimentalist claims to have raised the temperature of a small amount of water to 150°C by transferring heat from high pressure steam at 120°C. Is this a reasonable claim? Why? Assume no refrigerator or heat pump is used in the process.
> Reconsider Prob. 7–48. Using appropriate software, determine the power input required by the air conditioner to cool the house as a function of air conditioner EER ratings in the range 5 to 15. Discuss your results and include represent
> When a man returns to his well-sealed house on a summer day, he finds that the house is at 35°C. He turns on the air conditioner, which cools the entire house to 20°C in 30 min. If the COP of the air-conditioning system is 2.8, dete
> A vertical, frictionless piston–cylinder device contains a gas at 180 kPa absolute pressure. The atmospheric pressure outside is 100 kPa, and the piston area is 25 cm2. Determine the mass of the piston.
> A household refrigerator that has a power input of 450 W and a COP of 1.5 is to cool 5 large watermelons, 10 kg each, to 8°C. If the watermelons are initially at 28°C, determine how long it will take for the refrigerator to cool them. The watermelons ca
> An air conditioner removes heat steadily from a house at a rate of 750 kJ/min while drawing electric power at a rate of 5.25 kW. Determine (a) the COP of this air conditioner and (b) the rate of heat transfer to the outside air
> A heat pump with a COP of 1.4 is to produce a 100,000 Btu/h heating effect. How much power does this device require, in hp?
> A heat pump has a COP of 1.7. Determine the heat transferred to and from this heat pump when 50 kJ of work is supplied.
> Determine the COP of a heat pump that supplies energy to a house at a rate of 8000 kJ/h for each kW of electric power it draws. Also, determine the rate of energy absorption from the outdoor air.
> Determine the COP of a refrigerator that removes heat from the food compartment at a rate of 5040 kJ/h for each kW of power it consumes. Also, determine the rate of heat rejection to the outside air.
> A food refrigerator is to provide a 15,000-kJ/h cooling effect while rejecting 22,000 kJ/h of heat. Calculate the COP of this refrigerator.
> An automotive air conditioner produces a 1-kW cooling effect while consuming 0.75 kW of power. What is the rate at which heat is rejected from this air conditioner?
> Describe an imaginary process that satisfies the second law but violates the first law of thermodynamics.
> A food freezer is to produce a 5-kW cooling effect, and its COP is 1.3. How many kW of power will this refrigerator require for operation?
> The average temperature of the atmosphere in the world is approximated as a function of altitude by the relation Tatm = 288.15 − 6.5z where Tatm is the temperature of the atmosphere in K and z is the altitude in km with z = 0 at sea level. Determine the
> Show that the Kelvin–Planck and the Clausius expressions of the second law are equivalent.
> What is the Clausius expression of the second law of thermodynamics?
> A heat pump is a device that absorbs energy from the cold outdoor air and transfers it to the warmer indoors. Is this a violation of the second law of thermodynamics? Explain.
> In a refrigerator, heat is transferred from a lower temperature medium (the refrigerated space) to a higher temperature one (the kitchen air). Is this a violation of the second law of thermodynamics? Explain.
> A refrigerator has a COP of 1.5. That is, the refrigerator removes 1.5 kWh of energy from the refrigerated space for each 1 kWh of electricity it consumes. Is this a violation of the first law of thermodynamics? Explain.

