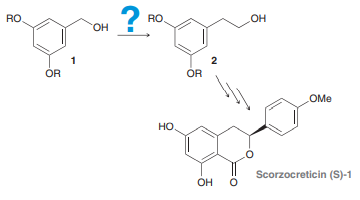
Question: Scorzocreticin (S)-1 was isolated from a
Scorzocreticin (S)-1 was isolated from a plant that is used for cooking traditional meat dishes on the Greek island of Crete. As part of a recent synthesis of scorzocreticin (S)-1, compound 1 was converted into compound 2. Identify a synthetic route for converting 1 into 2, knowing that it involves a Wittig reaction.
Transcribed Image Text:
?: RO RO OH 1 2 OR OR OMe HO Scorzocreticin (S)-1 OH
> Identify reagents that can be used to achieve the following transformation: OH
> Predict the major product(s) for each of the following reactions: 1) xs LIAIH, 2) H,0 ? 1) xs PhMgBr 2) H,0 1) LIAI(OR),H ? CI 2) EIMgBr 3) Н,О (b) (c) OH N-H "CI 1) El,CuLi ? CI 2) LIAIH, 3) H,0 Pyridine (two equivalents) (e) (f)
> Chlorocatechelin A was isolated from a bacterial culture, but it had to be synthesized in the laboratory in order to obtain enough pure material for the study of its antimicrobial activity. Shown below is the first step of a 15-step synthesis of chloroca
> Propose a plausible mechanism for each of the following transformations. These reactions will all appear later in this chapter, so practicing their mechanisms now will serve as preparation for the rest of this chapter: NH, O Na NH2 (a) (Ь) MEOH (c) H
> Draw resonance structures for the conjugate base that is produced when each of the following compounds is treated with sodium ethoxide: CN OEt (a) OEt (b) (c)
> Draw a structure for each of the following compounds: a. Dimethyl oxalate b. Phenyl cyclopentanecarboxylate c. N-Methylpropionamide d. Propionyl chloride
> Provide a name for each of the following compounds: (b) (c) (d) (f) (h) (1)
> Identify the reagents you would use to achieve each of the following transformations: Br HO. HO. (b)
> Identify the reagents you would use to perform the following transformations: a. Ethanol → Acetic acid b. Toluene → Benzoic acid c. Benzene → Benzoic acid d. 1-Bromobutane → Pentanoic acid e. Ethylbenzene → Benzoic acid f. Bromocyclohexane → Cycloh
> Rank each set of compounds in order of increasing acidity: a. 2,4-Dichlorobutyric acid 2,3-Dichlorobutyric acid 3,4-Dimethylbutyric acid b. 3-Bromopropionic acid 2,2-Dibromopropionic acid 3,3-Dibromopropionic acid
> Acetic acid was dissolved in a solution buffered to a pH of 5.76. Determine the ratio of the concentrations of acetate ion and acetic acid in this solution. Which species predominates under these conditions?
> When formic acid is treated with potassium hydroxide (KOH), an acid-base reaction occurs, forming a carboxylate ion. Draw a mechanism for this reaction and identify the name of the carboxylate salt.
> Based on your answer to the previous question, would you expect meta-hydroxyacetophenone to be more or less acidic than para-hydroxyacetophenone? Explain your answer. но. но meta-Hydroxyacetophenone para-Hydroxyacetophenone
> Consider the structure of para-hydroxyacetophenone, which has a pKa value in the same range as a carboxylic acid, despite the fact that it lacks a COOH group. Offer an explanation for the acidity of para-hydroxyacetophenone. но para-Hydroxyacetopheno
> The following two compounds are constitutional isomers. Identify which of these is expected to be more acidic and explain your choice. OH H3C OH
> One of the compounds from the previous problem has a pKa < 10. Identify that compound and explain why it is so much more acidic than all of the other compounds.
> Provide an IUPAC name for each of the following compounds: он (а) (b)
> Draw the structure of each of the following compounds: a. Cyclobutanecarboxylic acid b. 3,3-Dichlorobutyric acid c. 3,3-Dimethylglutaric acid
> Provide both an IUPAC name and a common name for each of the following compounds: a. HO2C(CH2)3CO2H b. CH3(CH2)2CO2H c. C6H5CO2H d. HO2C(CH2)2CO2H e. CH3COOH f. HCO2H
> Each of the following compounds does not participate as a diene in a Diels–Alder reaction. Explain why in each case. (a) (b) (c) (d)
> After performing the reaction from Problem 16.37, the reaction flask is heated to 40ºC and two of the products become the major products. When the flask is then cooled to 0ºC, no change occurs in the product distribution. Explain why an increase in tempe
> Draw all products that are expected when 2-ethyl-3-methyl 1,3-cyclohexadiene is treated with HBr at room temperature, and show a mechanism of their formation.
> In each of the following pairs of compounds identify the compound that liberates the most heat upon hydrogenation: (a) (b)
> Treatment of 1,2-dibromocycloheptane with excess potassium tert-butoxide yields a product that absorbs UV light. Identify the product.
> Circle each compound that has a conjugated π system:
> Draw the structure of each of the following compounds: a. 1,4-Cyclohexadiene b. 1,3-Cyclohexadiene c. (Z)-1,3-Pentadiene d. (2Z,4E)-Hepta-2,4-diene e. 2,3-Dimethyl-1,3-butadiene
> Identify which of the following compounds are expected to have a pKa он H.
> For each of the following tasks, use the color wheel in Figure 16.37. a. Identify the color of a compound that absorbs orange light. b. Identify the color of a compound that absorbs blue-green light. c. Identify the color of a compound that absorbs or
> As seen at the end of Section 16.10, a deficiency in vitamin D can cause a bonesoftening disease called rickets, so many foods, including milk and cereals, are fortified with vitamin D. Our bodies synthesize vitamin D when our skin is exposed to sunlight
> Use Woodward–Fieser rules to estimate the expected λmax for each of the following compounds: (b) (c) (d)
> Draw a plausible mechanism for the following transformation: Нeat
> Predict the product for each of the following reactions: ? Heat (a) ? Heat (b) ? Heat (c) ? Heat (d)
> Consider the structure of cis-1,2-divinylcyclopropane: This compound is stable at low temperature but rearranges at room temperature to produce 1,4-cycloheptadiene. a. Draw a mechanism for this transformation. b. Using brackets and numbers, identify
> For each of the following reactions, use brackets and two numbers to identify the type of sigmatropic rearrangement taking place: Нeat (а) H H. Нeat, (b)
> Inthomycin C, a natural product produced by Streptomyces bacteria, is known to inhibit the production of cellulose, an important component of the cell wall of plants. A recent synthesis of inthomycin C involved a thermal electrocyclic ring-opening reacti
> Predict the product(s) for each of the following reactions: ? ? hv hv Heat (b) (c)
> Predict the major product(s) for each of the following thermal electrocyclic reactions: . ? . ? ? Heat Heat Heat (a)
> Diastereoselective transformations, including the one shown below, have been developed during efforts toward the syntheses of antifungal and antibacterial natural products. Disregarding stereochemistry, propose an efficient synthesis for this transformat
> Consider the following [4+4] cycloaddition process. Would you expect this process to occur through a thermal or photochemical pathway? Justify your answer with MO theory.
> Predict the regiochemical outcome (major product) for each of the following Diels–Alder reactions: OMe .CN сно ? ? + (a) (Б) Мео CO,.Et EtO Meo сно ? -? CHO (c) (d) -?
> Treatment of the following ketone with LiAlH4 affords two products, A and B. Compound B has the molecular formula C8H14O and exhibits strong signals at 3305 cm−1 (broad) and 2117 cm−1 in its IR spectrum: a. Using the
> During a recent synthesis of hispidospermidin, a fungal isolate and an inhibitor of phospholipase C, the investigators employed a novel, aliphatic Friedel–Crafts acylation. The following acid chloride was treated with a Lewis acid, affo
> A convenient method for achieving the transformation below involves treatment of the ketone with Wittig reagent 1 followed by acid-catalyzed hydration: a. Predict the product of the Wittig reaction. b. Propose a plausible mechanism for the acid-catalyz
> During a recent total synthesis of englerin A, a potent cytotoxic natural product isolated from the stem bark of a Tanzanian plant, the investigators observed the following (unusual) basecatalyzed ring contraction. Propose a plausible mechanism for this
> During recent studies to determine the absolute stereochemistry of a bromohydrin, the investigators observed an unexpected skeletal rearrangement. Provide a plausible mechanism for the formation of epoxide 2 from bromohydrin 1. Br HO. NaH H. H 2
> The following two-step rearrangement was the cornerstone of the first stereoselective total synthesis of quadrone, a biologically active natural product isolated from an Aspergillus fungus. Propose a plausible mechanism for this transformation. OMs 1
> Prostaglandins (Section 26.7) are a group of structurally related compounds that function as biochemical regulators and exhibit a wide array of activity, including the regulation of blood pressure, blood clotting, gastric secretions, inflammation, kidney
> (−)-Spongidepsin is a marine natural product that is toxic to some cancer cells. During a synthesis of (−)-spongidepsin, compound 1 was converted into compound 2. Propose a synthesis for the conversion of 1 into 2.
> 46. Which monomer can be used to prepare the following polymer under acidic conditions? 47. Which monomers are formed when the following polymer undergoes acid-catalyzed hydrolysis? 48. Which polymer is formed by the condensation of these two monomers
> The plant Thapsia villosa is so poisonous that it is known as the deadly carrot! Despite the toxicity of this herb, its historical use in traditional medicine makes its extracted compounds attractive synthetic targets. A variety of compounds, called thap
> The m- and p-substituted methyl benzoates listed in the table below were treated with NaOH in dioxane and water. The rate constants of saponification, k, are also listed in the table. Provide structural explanations for the trend observed in the rate con
> Compound 3 (below) was used as an intermediate in a recently reported synthesis of the γ-secretase inhibitor BMS-708163. Compound 3 was made from compound 1 via the two-step process shown. a. Compound 1 cannot be directly converted into comp
> Allylic bromide 2 was recently used as a key fragment in progress toward the total synthesis of an analogue of amphidinolide N, a potent cytotoxic agent isolated from the marine organism Amphidinium sp. Propose an efficient synthesis of bromide 2 from ca
> Steroidal derivatives are important pharmacologically and biologically, and they continue to be a popular backbone in the search for new biologically active steroid hormone analogues. Propose a possible three-step synthesis for the conversion of steroid
> Aminotetralins are a class of compounds currently being studied for their promise as antidepressant drugs. The following conversion was employed during a mechanistic study associated with a synthetic route for preparing aminotetralin drugs. Propose a thr
> Olefin metathesis (2005 Nobel Prize) using the Grubbs catalyst has emerged as one of the most important tools for the organic chemist (e.g., 2 to 3). Metathesis substrate 2 can be prepared from 1 in three steps, shown below. Show the product of each synt
> Dimethylformamide (DMF) is a common solvent: a. The 1 H NMR spectrum of DMF exhibits three signals. Upon treatment with excess LiAlH4 followed by water, DMF is converted into a new compound that exhibits only one signal in its 1 H NMR spectrum. Explain.
> N-Acetylazoles undergo hydrolysis more readily than regular amides. Suggest a reason for the enhanced reactivity of N-acetylazoles toward nucleophilic acyl substitution. N- R. N-Acetylazole
> Propose an efficient synthesis for each of the following transformations: H. OH (а) (b) CI (c) (d) OH но. (e) (f)
> Propose a mechanism for the following transformation, and explain how you could use an isotopic labeling experiment to verify your proposed mechanism: [H,SO,] R но но
> A compound with the molecular formula C8H8O3 exhibits the following IR, 1H NMR, and 13C NMR spectra. Deduce the structure of this compound. Proton NMR 3 12 11 10 8 7 6 5 Chemical Shift (ppm) Carbon NMR 139.4 -118.3 128.0 64.1 172.8 -167.9 180 160 14
> Describe how you could use NMR spectroscopy to distinguish between benzoyl chloride and para-chlorobenzaldehyde.
> Describe how you could use IR spectroscopy to distinguish between ethyl acetate and butyric acid.
> A compound with the molecular formula C10H10O4 exhibits only two signals in its 1H NMR spectrum: a singlet at 4.0 (I = 3H) and a singlet at 8.1 (I = 2H). Identify the structure of this compound.
> Propose an efficient synthesis for each of the following transformations: (a) Br (b) (c) он CN NH (d) (e) Br H- (f) (g) - (h)
> Consider the three constitutional isomers of dioxane (C4H8O2): One of these constitutional isomers is stable under basic conditions as well as mildly acidic conditions and is therefore used as a common solvent. Another isomer is only stable under basic
> When cyclohexanone is treated with H2O, an equilibrium is established between cyclohexanone and its hydrate. This equilibrium greatly favors the ketone, and only trace amounts of the hydrate can be detected. In contrast, when cyclopropanone is treated wi
> Draw a plausible mechanism for the following transformation: NH2 `NH,
> Propose an efficient synthesis for each of the following transformations: (a) Meo OMe Br Br (b) (c)
> The following reaction utilizes a Michael addition, followed by a Claisen-type reaction to make chiral diketoester 3 as part of the total synthesis of dynemycin A, a natural product with promising anti-tumor activity in several human tumor cell lines. a
> The compound below is believed to be a wasp pheromone. Draw the major product formed when this compound is hydrolyzed in aqueous acid:
> Propose an efficient synthesis for each of the following transformations: (а) (b)
> Using ethanol as your only source of carbon atoms, design a synthesis for the following compound:
> Identify the starting materials needed to make each of the following acetals: x .C OEt (a) (b) (c) o
> Predict the major product(s) for each of the following reactions: CH3 NH2 ? (a) ? 1) PhMgBr 2) H20 (b) ? CH;COH (c) CH,CO,H ? (d) ? -NH2 (-H20) (e)
> Draw a plausible mechanism for each of the following transformations: H,0. (а) H. H,0", H,N. H. (b) [H1 H. но (c) он I-Z
> Identify all of the expected products when the compound below is treated with aqueous acid: ? Excess H,0+ N-
> Predict the major product(s) obtained when each of the following compounds undergoes hydrolysis in the presence of H3O+: (a) (b) (c) (d)
> Identify the reactants that you would use to make each of the following enamines: (a) (b) (с)
> Identify the reactants that you would use to make each of the following imines: (a) (b) (c)
> Using 1-propanol as your only source of carbon, propose an efficient synthesis for each of the following compounds: он H. но. OH (а) (b) (с) н H.
> Glutaraldehyde is a germicidal agent that is sometimes used to sterilize medical equipment too sensitive to be heated in an autoclave. In mildly acidic conditions, glutaraldehyde exists in a cyclic form (below right). Draw a plausible mechanism for this
> Starting with cyclopentanone and using any other reagents of your choosing, identify how you would prepare each of the following compounds: но соон (a) (b) (c) (d)
> Treatment of catechol with formaldehyde in the presence of an acid catalyst produces a compound with the molecular formula C7H6O2. Draw the structure of this product. он HO. Catechol
> Propose a plausible mechanism for the following transformation: но H. EIOH
> Predict the major product(s) from the treatment of acetone with the following: a. [H+], NH3, (−H2O) b. [H+], CH3NH2, (−H2O) c. [H+], excess EtOH, (−H2O) d. [H+], (CH3)2NH, (−H2O) e. [H+], NH2NH2, (−H2O) f. [H+], NH2OH, (−H2O) g. NaBH4, MeOH h. RC
> You are working in a laboratory, and you are given the task of converting cyclopentene into 1,5-pentanediol. Your first thought is simply to perform an ozonolysis followed by reduction with LiAlH4, but your lab is not equipped for an ozonolysis reaction.
> Choose a Grignard reagent and a ketone that can be used to produce each of the following compounds: a. 3-Methyl-3-pentanol b. 1-Ethylcyclohexanol c. Triphenylmethanol d. 5-Phenyl-5-nonanol
> Draw the structure of the alkyl halide needed to prepare each of the following Wittig reagents and then determine which Wittig reagent will be the more difficult to prepare. Explain your choice. Ph. Ph Ph, Ph-P= Ph Ph-P H Ph
> Draw the major product of each Wittig reaction below: Ph Ph. Ph-P= Ph (a) Ph, Ph-P= Ph ? Ph, H. (b) Ph
> For each pair of the following compounds, identify which compound would be expected to react more rapidly with a nucleophile: H. (b) F.C CF3 H,C CH, (a)
> Using cyclopentanone as your starting material and using any other reagents of your choice, propose an efficient synthesis for each of the following compounds: он (а) (Ь) (с)
> Draw all constitutionally isomeric ketones with the molecular formula C6H12O and provide a systematic (IUPAC) name for each isomer.
> Draw all constitutionally isomeric aldehydes with the molecular formula C5H10O and provide a systematic (IUPAC) name for each isomer. Which of these isomers possesses a chiral center?
> Draw all constitutionally isomeric aldehydes with the molecular formula C4H8O and provide a systematic (IUPAC) name for each isomer.
> Provide a systematic (IUPAC) name for the compound below. Be careful: This compound has two chiral centers (can you find them?).
> Draw the structure for each compound below: a. Propanedial b. 4-Phenylbutanal c. (S)-3-Phenylbutanal d. 3,3,5,5-Tetramethyl-4-heptanone e. (R)-3-Hydroxypentanal f. meta-Hydroxyacetophenone g. 2,4,6-Trinitrobenzaldehyde h. Tribromoacetaldehyde i.
> Provide a systematic (IUPAC) name for each of the following compounds: (a) (b) (c) H. (d)
> Compound A has the molecular formula C10H10O and exhibits a strong signal at 1720 cm−1 in its IR spectrum. Treatment with 1,2-ethanedithiol followed by Raney nickel affords the product shown. Identify the structure of compound A. 1)
> The anti-tumor compound maytansine was originally isolated from the Ethiopian shrub Maytenus serrata. Development of a multistep synthesis of maytansine involved compound 1 as a key precursor. Propose an efficient synthesis of 1 starting with acetylene,

