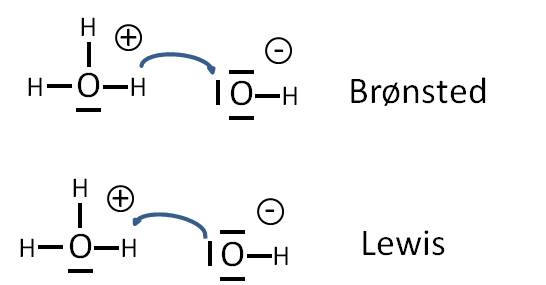
Definition of Lewis Base
A Lewis base is defined as a substance that can donate a pair of electrons to form a covalent bond. Hence, they are known to be the electron pair donors. In the Lewis acid-base reaction, the pair of electrons are transferred from the base to an acid.
According to Lewis and Bronsted Lowry, the hydrogen ion (H+) is said to be acid as it is deficient in electrons and therefore accepts a pair of electrons. Whereas, ammonia (NH3) is known to be a Lewis base as the nitrogen atom has a lone pair of electrons that can be donated to acid to form a covalent bond.
View More Organic Chemistry Definitions
Related Questions of Organic Chemistry
Draw and name four terminal alkynes with the molecular formula C6H10.
Report the result of the following addition to the proper number of
Scorzocreticin (S)-1 was isolated from a plant that is
Write an equation for the proton transfer reaction that occurs when each
87. What is the major product of this reaction?
Propose a stepwise mechanism for the following transformation: /
Identify the sole product of the following reaction: /
Draw the condensation product that is expected when each of the following
Draw a mechanism for each of the following transformations: /
Draw each of the following using condensed formulas and line formulas:
Show All